Blog Authors:
RK Neelakandan - Health Software Quality and Solutions Lead
Teginder Singh - Global Head of Regulatory and Safety Operations
In the highly regulated Life Sciences sector, the pace of technological innovation often outstrips the pace of compliance. While engineering teams leverage cloud-native infrastructure and Generative AI to accelerate drug discovery and clinical analysis, quality assurance processes frequently remain tethered to static, document-centric validation methodologies.
This dissonance creates a phenomenon we call "Validation Debt"—the accumulation of manual verification tasks that slow down release cycles and inhibit the adoption of modern technologies. For organizations aiming to modernize their GxP (Good Practice) workloads, the challenge is no longer just about maintaining compliance; it is about harmonizing regulatory rigor with the velocity of modern DevOps.
The traditional "snapshot" approach to Computer System Validation (CSV)—where a system is validated at a single point in time—is becoming increasingly difficult to reconcile with ephemeral cloud infrastructure and non-deterministic AI models. To bridge this gap, forward-thinking organizations are shifting toward Model-Driven Continuous Validation.
This article outlines a reference architecture for implementing this approach on Google Cloud, transforming validation from a periodic gatekeeping exercise into an "always-on" assurance function.
The Strategic Shift: From Periodic to Continuous
The FDA’s shift toward Computer Software Assurance (CSA) emphasizes critical thinking and risk-based testing over burdensome documentation. However, implementing CSA requires a technical architecture capable of proving the system’s "validated state" continuously.
In a modern cloud environment, relying on manual scripts and periodic audits introduces significant blind spots:
-
Dynamic Infrastructure Provisioning & drift: In modern environments, resources are often provisioned dynamically or subject to configuration drift. A static validation report generated at the time of deployment cannot guarantee the current, ongoing state of the infrastructure.
-
Probabilistic AI Behaviors: As Life Sciences organizations integrate Large Language Models (LLMs) for tasks like summarizing notes, standard binary (pass/fail) testing becomes insufficient. These systems require probabilistic evaluation to ensure safety and accuracy over time.
To address these complexities, we propose a dual-pathway architecture. This framework relies on a machine-readable "Compliance Model"—a codified set of regulatory requirements—that orchestrates validation across two parallel streams: a Proactive Digital Twin for pre-production stress testing, and a Reactive Sentinel for real-time production monitoring.
Architecture: The Google Cloud Framework for Continuous GxP
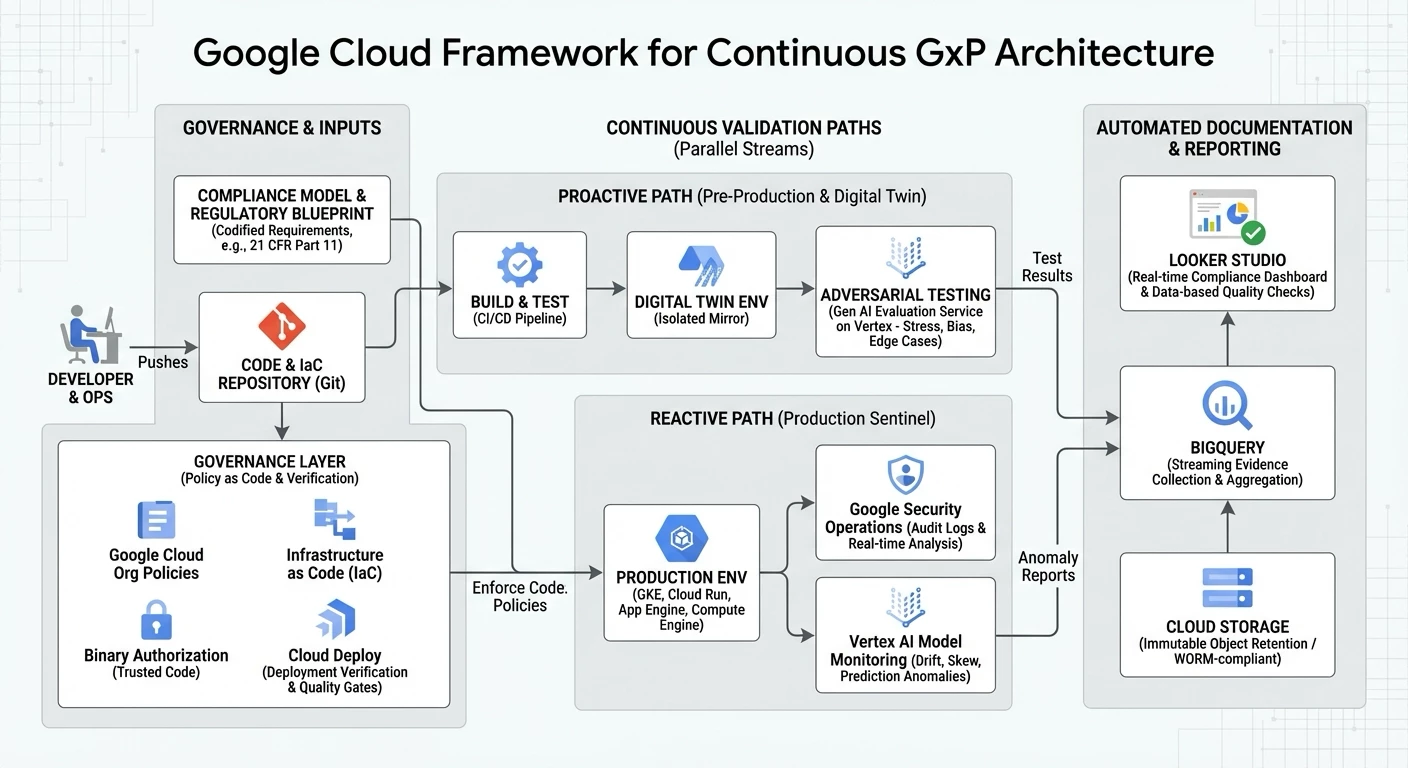
By leveraging Google Cloud’s integrated data and security portfolio, organizations can construct a Framework for Continuous GxP that enforces governance without impeding development velocity. The reference architecture below illustrates how the Compliance Model orchestrates validation across the Proactive and Reactive paths:
1. Governance: Codifying the "Regulatory Blueprint"
The foundation of continuous validation is the translation of written requirements (e.g., 21 CFR Part 11, GAMP 5) into executable code.
Organizations can establish a "Regulatory Blueprint" across diverse environments—whether utilizing Google Kubernetes Engine (GKE), serverless architectures like Cloud Run and App Engine, or Compute Engine VMs. This involves hard-coding GxP guardrails—such as encryption standards, residency restrictions, and access controls—using Google Cloud Organization Policies and Infrastructure as Code (IaC).
To enforce these policies, organizations can secure their software supply chain with Binary Authorization, ensuring only trusted and verified code is deployed. Furthermore, utilizing Cloud Deploy with Deployment Verification integrates continuous delivery with automated, pre-configured quality gates. By treating policy as code, the platform itself acts as the primary gatekeeper, automatically rejecting any deployment that deviates from the compliance model before it reaches production.
2. Verification: Adversarial Testing in the Digital Twin
For critical GxP workloads, particularly those involving AI, standard functional testing is often insufficient.
In the Proactive Path, code is deployed to a high-fidelity Digital Twin—an isolated environment that mirrors production. Here, we introduce Gen AI Evaluation Service on Vertex to perform adversarial "red teaming." Rather than simply checking for expected outputs, the system actively challenges the application or AI model with edge cases, noise, and bias tests. This rigorous stress-testing provides mathematical evidence of model robustness, satisfying the "intended use" validation requirements for complex, non-deterministic systems.
Furthermore, the Digital Twin can serve as an ongoing control for production integrity. By feeding live data from the production process into the twin (often called "shadow testing"), organizations can compare outputs side-by-side to detect subtle variations, bridging the gap between proactive testing and real-time monitoring.
3. Observability: The Production Sentinel
Validation does not end at deployment. The Reactive Path ensures that the system remains in its validated state during operation.
By ingesting audit logs into Google Security Operations and leveraging Vertex AI Model Monitoring, organizations can deploy a "Production Sentinel." This system monitors for behavioral drift, data skew, prediction drift, and infrastructure anomalies in real-time. For example, if an administrator executes a database change that bypasses the established change control workflow, or an AI model begins to drift outside acceptable accuracy parameters, the Sentinel correlates these events against the compliance model and flags the deviation immediately. This shifts the posture from "auditing the past" to "monitoring the present."
4. Documentation: The Automated Scribe
Finally, to reduce the administrative burden of validation, the architecture automates evidence collection.
All test results from the Digital Twin and anomaly reports from the Production Sentinel are streamed into BigQuery. To meet data integrity standards (ALCOA+), Cloud Storage Object Retention can be applied to ensure this record is immutable (WORM-compliant). Visualization tools like Looker Studio then provide a real-time compliance dashboard, where data-based quality checks can also be automated, replacing static binders with a dynamic view of the system’s health.
Furthermore, rather than keeping this data siloed, these automated validation events and anomaly reports can be routed via Pub/Sub to integrate directly with enterprise Quality Management Systems (QMS) like Veeva Vault via standard APIs, ensuring a single source of truth for all quality records.
The Takeaway
Modernizing validation is a strategic imperative for Life Sciences organizations seeking to leverage the full potential of cloud and AI technologies. By adopting a Model-Driven Continuous Validation approach, leaders can achieve three critical business outcomes:
-
Accelerated Time-to-Market: By automating the verification of infrastructure and code, organizations reduce the "validation lag" that traditionally delays releases.
-
Enhanced Risk Posture: Continuous monitoring and adversarial testing provide a higher level of assurance than periodic manual checks, particularly for AI-driven workloads.
-
Regulatory Readiness: An automated, immutable chain of custody for evidence simplifies audits and aligns with modern regulatory guidance like CSA.
As the industry evolves, the ability to validate continuously will become a defining characteristic of agile, compliant, and innovative Life Sciences enterprises.
Ready to build Google Cloud Framework for Continuous GxP for your life sciences needs? Explore our Life Sciences solutions page or contact our sales team to get started.



